Trending...
- Report: Chicago's Safety Plan Showing Promising Results in Key Neighborhoods
- CCHR Warns: Psychiatric Diagnoses Without Biological Proof Now Used to Justify Euthanasia
- Scoop Social Co. Launches a New Era of Mobile Hospitality — One Truck, Two Experiences
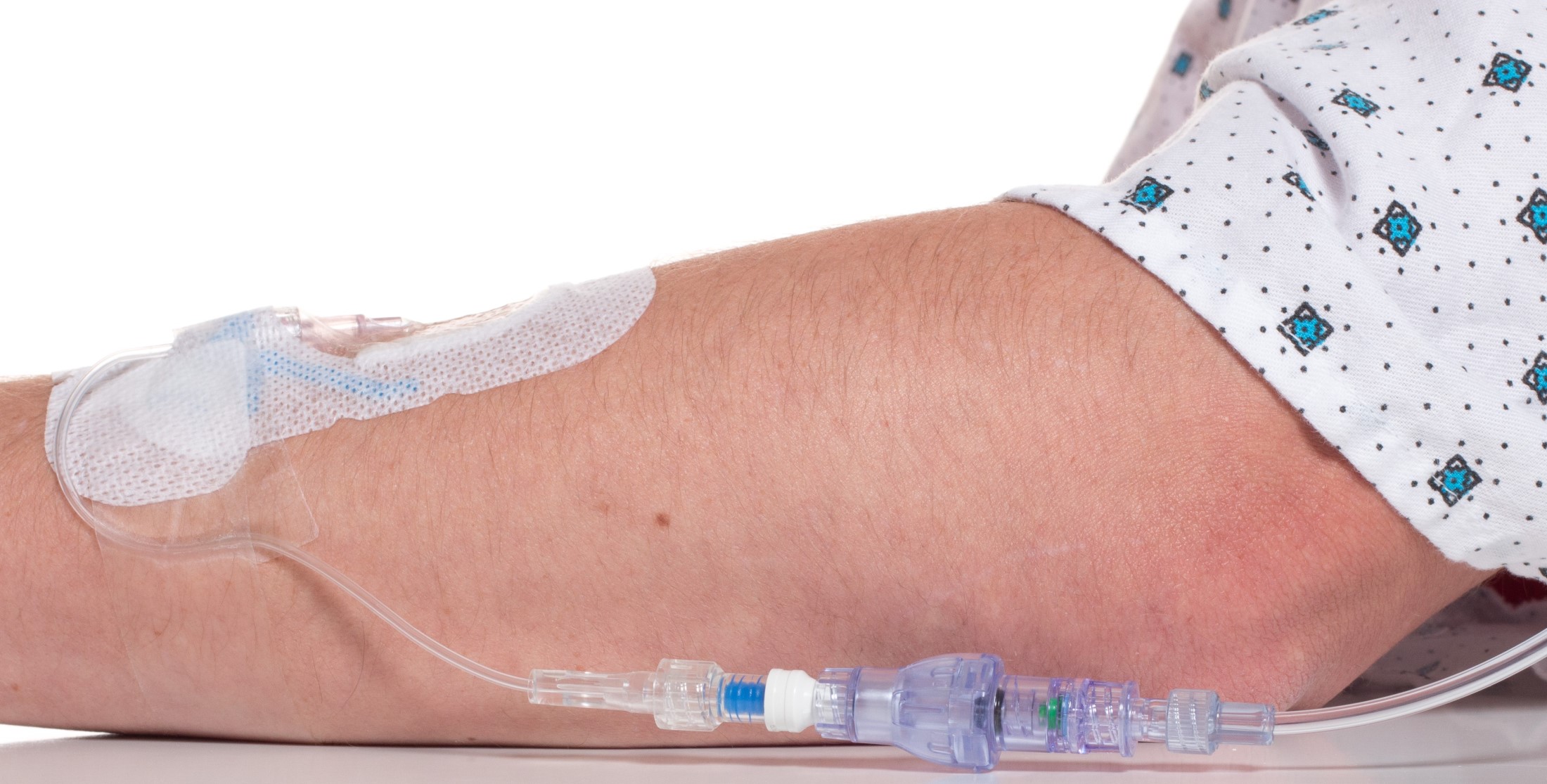
Lineus Medical Creates New Class of Medical Device Intended to Reduce Complications in Peripheral IV Lines
FAYETTEVILLE, Ark. - illiNews -- The US Food and Drug Administration has cleared for sale Lineus Medical's first medical device, SafeBreak Vascular, that reduces mechanical complications requiring IV restarts in peripheral IV lines when excessive tension (greater than 4 lbs. of force) is placed on the IV line. Tugs or pulls on IV lines are a common occurrence and part of every patient's time in the hospital. The current method for securing IV catheters includes the use of adhesive dressings and securement devices. All current methods focus on securing the line to the patient's skin and these solutions fail when the IV tubing is subjected to large pull forces. SafeBreak Vascular works differently than current methods and is part of a new class of devices called Force-Activated Separation Devices. Force-Activated Separation Devices separate to remove the damaging force from the IV line and prevent the loss of blood and medication.
SafeBreak Vascular is the first ever Force-Activated Separation Device cleared to be sold in the United States through the FDA's De Novo review process. Upon separation, a valve in each end of SafeBreak closes. On the patient side, the IV is left intact and the valve closes and prevents the patient from bleeding. On the IV pump side, the valve closes and stops the pump from pumping medicine onto the floor or in the patient's bed. When the fluid flow from the IV pump is stopped, it causes the pump to alarm, letting the medical team know they need to address the situation. The separated SafeBreak Vascular can be discarded and a new one installed in the line in only a few minutes. The patient's medical treatment can resume by restarting the IV infusion without the need for a new catheter or additional needle sticks.
More on illi News
The clinical literature shows that over 235 million peripheral IV lines are placed each year in the United States and that these IV lines fail before their intended end of use a staggering 46% of the time on average. The cost of an IV restart is approximately $50, and IV restarts are estimated to cost the US healthcare system over $5 billion annually.
Lineus Medical was founded in 2015 to develop and bring SafeBreak Vascular to market. "Achieving FDA De Novo Designation as a Class II device is no doubt the biggest milestone to date for our company. We're ecstatic about reaching this inflection point and eager to start executing on our product launch plan. It took an incredible amount of perseverance, a lot of investment, and a talented team to overcome the challenges we faced along the way.", said Lineus Medical's founder and Chief Technology Officer, Spencer Jones.
Vance Clement, CEO of Lineus Medical, stated, "We want to thank our investors for their continued support and especially through the Covid-19 pandemic. A randomized clinical trial that was completed in 2021 showed the device worked as advertised, eliminating spills, and saving nurses a great deal of time. The study most importantly showed a 44% reduction of peripheral IV complications requiring an IV restart in the SafeBreak Vascular group when compared to the control group. Now we are excited to get the product into the hands of clinicians everywhere so that we can start to positively impact a lot more patient's lives."
More on illi News
SafeBreak Vascular is distributed on behalf of Lineus Medical by MediCore of Nashville, TN along with multiple other independent sub-distributors across the entire United States. MediCore distributes other vascular access products to over 1100 US hospitals.
Spencer Jones said, "As a nurse myself, I know that during most shifts nurses are just worried about keeping their heads above water. There's too much to do and never enough time. Empowering nurses is core to our company's mission, and I'm thrilled that we're finally able to offer nurses a device that saves them time, helps reduce stressful IV restarts, and helps them start saving lines. Our team is looking forward to an exciting launch!"
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical tubing may be reduced as force-activated separation devices separate to protect the patient. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Twitter or LinkedIn.
MKG-0095 06/21 Rev 00
SafeBreak Vascular is the first ever Force-Activated Separation Device cleared to be sold in the United States through the FDA's De Novo review process. Upon separation, a valve in each end of SafeBreak closes. On the patient side, the IV is left intact and the valve closes and prevents the patient from bleeding. On the IV pump side, the valve closes and stops the pump from pumping medicine onto the floor or in the patient's bed. When the fluid flow from the IV pump is stopped, it causes the pump to alarm, letting the medical team know they need to address the situation. The separated SafeBreak Vascular can be discarded and a new one installed in the line in only a few minutes. The patient's medical treatment can resume by restarting the IV infusion without the need for a new catheter or additional needle sticks.
More on illi News
- USA Best Book Awards Finalist What Love Leaves Behind Releases March 24
- City Of Chicago Challenges Unlawful Rescission of Landmark 2009 Greenhouse Gas Endangerment Finding
- Vanguard's Happy Campers Headed to Sphero World Championship
- Inkdnylon Custom Apparel Launches Cost-Saving System for Promotional Products and Custom Apparel in Chicago
- ENTOUCH Named Finalist for 2026 North American Inspiring Workplaces Awards
The clinical literature shows that over 235 million peripheral IV lines are placed each year in the United States and that these IV lines fail before their intended end of use a staggering 46% of the time on average. The cost of an IV restart is approximately $50, and IV restarts are estimated to cost the US healthcare system over $5 billion annually.
Lineus Medical was founded in 2015 to develop and bring SafeBreak Vascular to market. "Achieving FDA De Novo Designation as a Class II device is no doubt the biggest milestone to date for our company. We're ecstatic about reaching this inflection point and eager to start executing on our product launch plan. It took an incredible amount of perseverance, a lot of investment, and a talented team to overcome the challenges we faced along the way.", said Lineus Medical's founder and Chief Technology Officer, Spencer Jones.
Vance Clement, CEO of Lineus Medical, stated, "We want to thank our investors for their continued support and especially through the Covid-19 pandemic. A randomized clinical trial that was completed in 2021 showed the device worked as advertised, eliminating spills, and saving nurses a great deal of time. The study most importantly showed a 44% reduction of peripheral IV complications requiring an IV restart in the SafeBreak Vascular group when compared to the control group. Now we are excited to get the product into the hands of clinicians everywhere so that we can start to positively impact a lot more patient's lives."
More on illi News
- Keyrenter Property Management Naperville Announces Grand Opening
- Cleveland County Goat Farm NC Kikos Featured in "Feature Farmer Friday" Documentary
- Registration for Future of Commerce 2026 is Now Open!
- Tony Grundler Introduces Artificial Intelligence V.S. Avatar-Ian's
- Hollywood's Elite Gather at the Annual WOW Creations Oscars Gifting Suite at the Universal Hilton
SafeBreak Vascular is distributed on behalf of Lineus Medical by MediCore of Nashville, TN along with multiple other independent sub-distributors across the entire United States. MediCore distributes other vascular access products to over 1100 US hospitals.
Spencer Jones said, "As a nurse myself, I know that during most shifts nurses are just worried about keeping their heads above water. There's too much to do and never enough time. Empowering nurses is core to our company's mission, and I'm thrilled that we're finally able to offer nurses a device that saves them time, helps reduce stressful IV restarts, and helps them start saving lines. Our team is looking forward to an exciting launch!"
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical tubing may be reduced as force-activated separation devices separate to protect the patient. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Twitter or LinkedIn.
MKG-0095 06/21 Rev 00
Source: Lineus Medical
Filed Under: Health
0 Comments
Latest on illi News
- Chicago: Mayor Brandon Johnson Applauds Passage of Ald. Fuentes Led Measure Empowering COPA to Investigate Alleged Welcoming City Violations
- Chicago: Mayor Brandon Johnson, CFD Commissioner Nance-Holt Mourn the Passing of Firefighter-EMT Michael Altman
- Jackets for Jobs Hosts Smart & Sexy® Day Detroit for Women's History Month
- Klincher Live at Pop's Nightclub Brings High-Energy Rock Show to Sauget Illinois
- Jeron Electronic Systems, Inc. to Showcase Area of Rescue Pro-Alert™ 480 at ISC West 2026, Booth #6130
- A Year in Love by Michael G. D'Aversa Offers Daily Celebrations of Romance and Devotion
- Mayor Brandon Johnson and Chicago City Council Honor, Celebrate, And Memorialize the Life and Legacy of the Rev. Jesse Jackson, Extend Condolences to the Jackson Family
- Tint Academy Training in Dallas Texas: Learn Window Tint & PPF Installation
- $IBG accelerates toward transformative merger with BlockFuel as $6 million raise fuels dual-industry growth strategy: N A S D A Q: IBG
- High-Growth Power Infrastructure Play Targets AI Boom: 1606 Corp. Executes Aggressive Texas Expansion Strategy: 1606 Corp. (Stock Symbol: CBDW) $CBDW
- Accelerating the Transformation into a U.S. Nuclear Fuel Cycle Leader: Frontier Nuclear and Minerals Inc. (N A S D A Q: FNUC)
- Ozz Metals Ltd Secures 1-Tonne Gold Offtake Agreement
- Jet Set: The Ultimate Coachella Afterparty
- Heritage at Manalapan Introduces New Single-Family Home Community in One of Monmouth County's Most Desirable Locations
- Compliant Workspace announces partnership with Blackpoint Cyber
- Michigan Homeowners Urged to Act on Rising Basement Waterproofing Needs Amid Severe Flood
- Event Solutions Enters New Era: Announces New Leadership
- Carlsbad Hotel Named Best of La Quinta Award Winner
- Scoop Social Co. Launches a New Era of Mobile Hospitality — One Truck, Two Experiences
- Record Sales Growth After Strategic Acquisitions; New Distribution Agreements for Established Premium Cigar Supplier: Green Leaf Innovations $GRLF