Trending...
- Kettera Strategies Selects Bite Stream
- NYC Composer/Educator Launches Debut Children's Book to Fantastic Reviews
- Red5 Taps PubNub to Power the Next Era of Real-Time Interactive Streaming
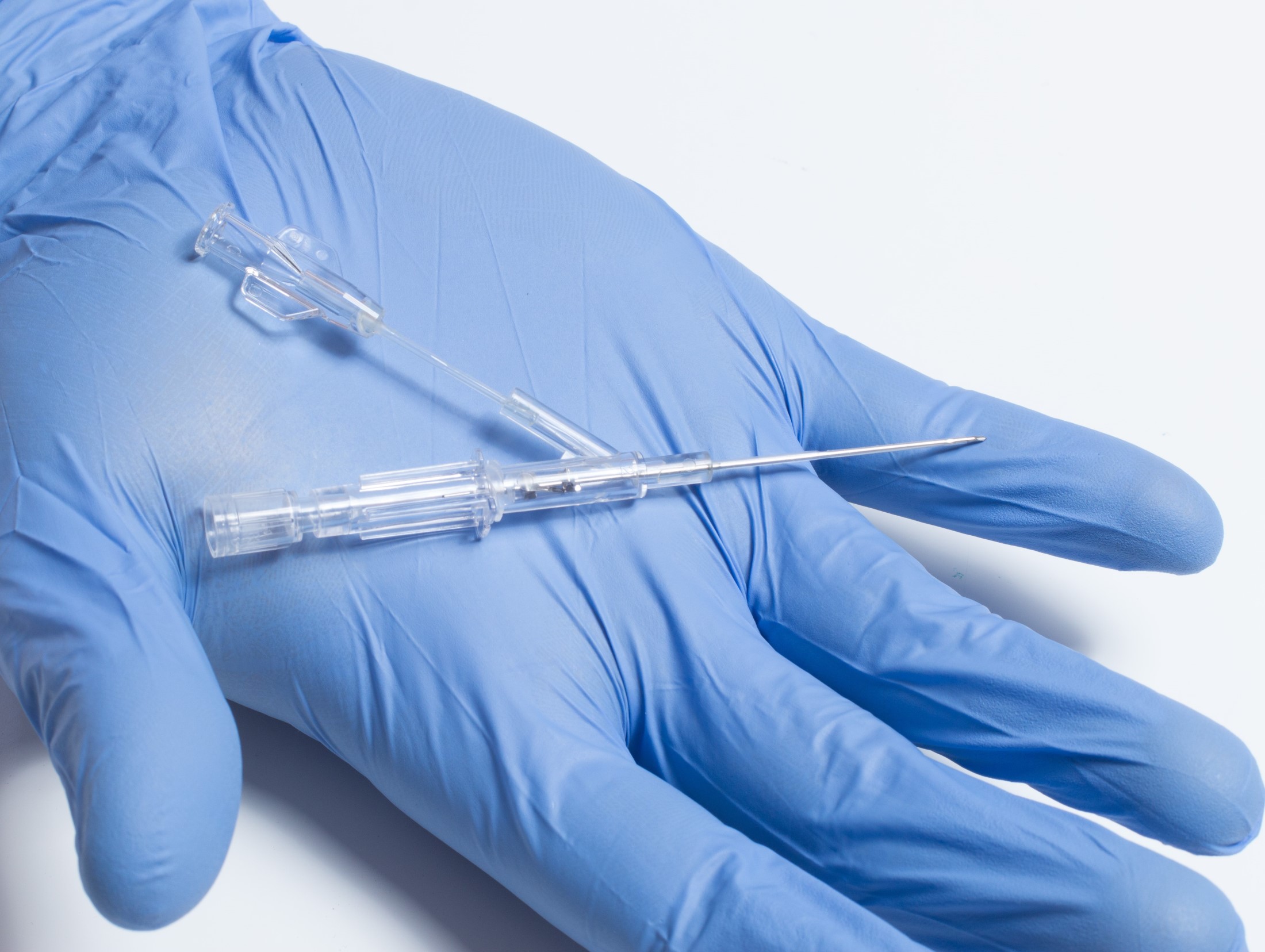
FAYETTEVILLE, Ark. & LITTLE ROCK, Ark. - illiNews -- Lineus Medical announces it has been awarded Technology Development Program (TDP) funding through the Arkansas Economic Development Commission (AEDC). The TDP provides royalty based financing for qualified science and technology projects with a potential for economic and employment growth in the state of Arkansas. The goal of the program is to assist in developing new technology-based products and processes. The $100K in funds will be used to further develop BVAD, Lineus Medical's patented, dual lumen, peripheral IV catheter.
This is the first time Lineus Medical has received TDP funding from the AEDC. Vance Clement, CEO of Lineus Medical stated, "The design enhancement we're investigating has the potential to reduce catheter occlusion, a common complication that impacts millions of IV catheters every year. Additionally, this design enhancement would give us a differentiated product offering for the massive single lumen peripheral IV catheter market. We are very grateful for the support we've received from the AEDC."
More on illi News
BVAD is the company's second product behind SafeBreak Vascular, the company's flagship product that received FDA clearance in May of 2021. BVAD is a peripheral IV catheter designed to dwell in the vein while facilitating pain free, uncontaminated blood samples through a secondary lumen dedicated to only blood draws. Lineus Medical believes the use of BVAD will likely reduce phlebotomy costs and make inpatient hospital stays more comfortable for diabetics and other patients that require frequent blood sampling.
Spencer Jones, Founder and Chief Technology Officer stated, "This TDP funding is critical because it allows BVAD development to move forward in a meaningful way. With these funds, we will be able to validate the safety and performance of a unique design enhancement against industry leading peripheral IV catheters."
The funding awarded through the TDP is non-dilutive and does not require matching funds from the company receiving the award. Royalties from sales generated from the developed technology will be collected by the AEDC in accordance with the royalty agreement reached by the AEDC and the award recipient.
More on illi News
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that can be adapted for use with a variety of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical line could be prevented with a device that breaks into two pieces to protect the patient's medical line. More information about Lineus Medical can be found at www.lineusmed.com. Follow Lineus Medical on LinkedIn or Twitter.
ABOUT THE AEDC
The mission of the Arkansas Economic Development Commission is to create economic opportunity by attracting higher paying jobs, expanding and diversifying Arkansas' state and local economies, increasing incomes and investment, and generating positive growth throughout Arkansas. The AEDC offers a number of programs, resources and incentives that serve businesses across the entire state. For more information on programs and incentives offered by AEDC, click this link.
This is the first time Lineus Medical has received TDP funding from the AEDC. Vance Clement, CEO of Lineus Medical stated, "The design enhancement we're investigating has the potential to reduce catheter occlusion, a common complication that impacts millions of IV catheters every year. Additionally, this design enhancement would give us a differentiated product offering for the massive single lumen peripheral IV catheter market. We are very grateful for the support we've received from the AEDC."
More on illi News
- The City of Naperville Launches 2026 National Community Survey
- NRx Pharmaceuticals Could Be on the Verge of a Breakout Year as AI, FDA Catalysts, and Mental Health Demand Converge
- DC Accounting Firm Offers Free Business CRM to Small Business Clients Alongside Weekly Bookkeeping Model
- CCHR: Psychiatric Drugs Fuel Rising Death Toll: National Adverse Drug Event Awareness Day Confronts America's Medication Crisis
- Explosive $10 Billion Counter-Drone Market with AI-Powered Defense Ecosystem: ZenaTech, Inc. (N A S D A Q: ZENA)
BVAD is the company's second product behind SafeBreak Vascular, the company's flagship product that received FDA clearance in May of 2021. BVAD is a peripheral IV catheter designed to dwell in the vein while facilitating pain free, uncontaminated blood samples through a secondary lumen dedicated to only blood draws. Lineus Medical believes the use of BVAD will likely reduce phlebotomy costs and make inpatient hospital stays more comfortable for diabetics and other patients that require frequent blood sampling.
Spencer Jones, Founder and Chief Technology Officer stated, "This TDP funding is critical because it allows BVAD development to move forward in a meaningful way. With these funds, we will be able to validate the safety and performance of a unique design enhancement against industry leading peripheral IV catheters."
The funding awarded through the TDP is non-dilutive and does not require matching funds from the company receiving the award. Royalties from sales generated from the developed technology will be collected by the AEDC in accordance with the royalty agreement reached by the AEDC and the award recipient.
More on illi News
- High-Value Execution Phase Begins: Bitcoin Bancorp Ignites Texas Rollout of Digital Asset ATM Network: Bitcoin Bancorp (Stock Symbol: BCBC) $BCBC
- Upgrade Kitchen Performance with SMEG Parts at HnK Parts for Ovens & Fridges
- PIXALPHA STUDIO Launches as Chicago's Premier Custom Web Design Agency
- UK Financial Ltd Tokenized LTNS 1, A $1.1 T Asset-Backed ERC-3643 Security Token with 11 On-Chain Contracts Verifying, Compliant Real-World Value
- SelfCare is now HealthCare across America
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that can be adapted for use with a variety of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical line could be prevented with a device that breaks into two pieces to protect the patient's medical line. More information about Lineus Medical can be found at www.lineusmed.com. Follow Lineus Medical on LinkedIn or Twitter.
ABOUT THE AEDC
The mission of the Arkansas Economic Development Commission is to create economic opportunity by attracting higher paying jobs, expanding and diversifying Arkansas' state and local economies, increasing incomes and investment, and generating positive growth throughout Arkansas. The AEDC offers a number of programs, resources and incentives that serve businesses across the entire state. For more information on programs and incentives offered by AEDC, click this link.
Source: Lineus Medical
0 Comments
Latest on illi News
- Chicago: Mayor Brandon Johnson Joins Goodman Theatre To Celebrate Opening of Theater of The Mind
- Chicago: Mayor Brandon Johnson Statement on Preliminary Ruling Restoring Red Line Extension Funding
- Kettera Strategies Selects Bite Stream
- Mark Dobosz Makes Donorassess.org Free To Every Nonprofit On The Planet
- Genpak Announces Closure of Utah Manufacturing Facility
- Vanguard Gifted Academy Students Join Millions Globally for World Maths Day 2026
- Newborn Care Network Introduces Clinical Standard to Bridge the Six-Week Postpartum Gap
- The AAA Metamorphosis: How Global Gaming Is Redefining Production Standards
- Monexplora Explains the Options Mechanics Behind March's Tech Selloff and VIX Surge
- Ill House District 83, Candidate, Loyola Grad Calls for Corrective Action Due to Gorman Murder
- Larry R. Wasion Highlights Jump Gate I: Time Chair. The Opening Novel in His Expansive Science Fiction Series
- Mayor Brandon Johnson Launches Repair Chicago Engagement Series and Survey to Inform City's Reparations Study
- New Book Reveals The Science Of Predictions
- Compliance Alert: Maryland, Texas Regulate Use of Artificial Intelligence in Utilization Reviews
- Elite Benefits Launches "Benefits Survival Guide" to Help Employers Control Costs and Uncertainty
- Colony Ridge Communities Celebrates Successful Soccer Season Kickoff with Families and Youth
- Pregis Launches Sharp™ Lynx 18 Automated Bagger: Unlocking Packing Efficiency in Space-Constrained Fulfillment Operations
- NYC Composer/Educator Launches Debut Children's Book to Fantastic Reviews
- EFA Announces 2026 Editorial Rate Chart
- Red5 Taps PubNub to Power the Next Era of Real-Time Interactive Streaming