Trending...
- Innovative Ear Muff Hat Combines Warmth and Convenience in One Winter Accessory
- New Portable Toilet Drain Adapter Eliminates Manual Waste Tank Emptying
- Kanguro Insurance Taps Paylode to Launch Best-in-Class Pet and Renters Insurance Rewards Experience
BOZEN, Italy - illiNews -- OncoBeta®, a leading medical device company specialising in innovative epidermal radioisotope therapies, has announced the expansion of Rhenium-SCT® (Skin Cancer Therapy) treatment services in Italy with the Bolzano Hospital the latest to come on board. This marks a significant advancement in the availability of non-melanoma skin cancer (NMSC) treatment options within the country.
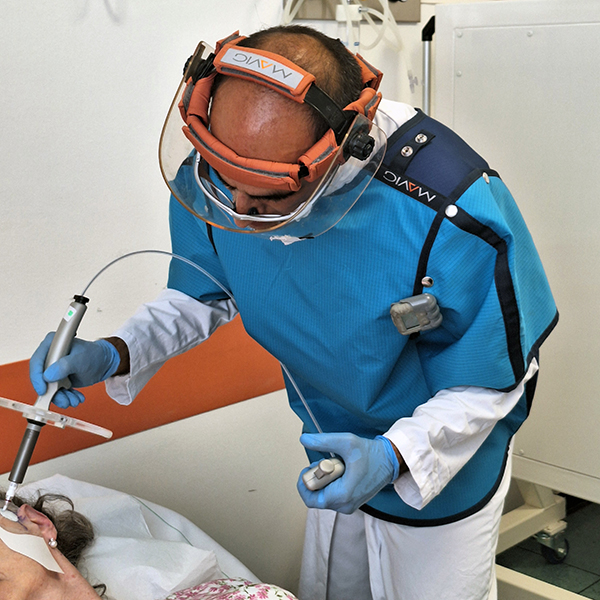
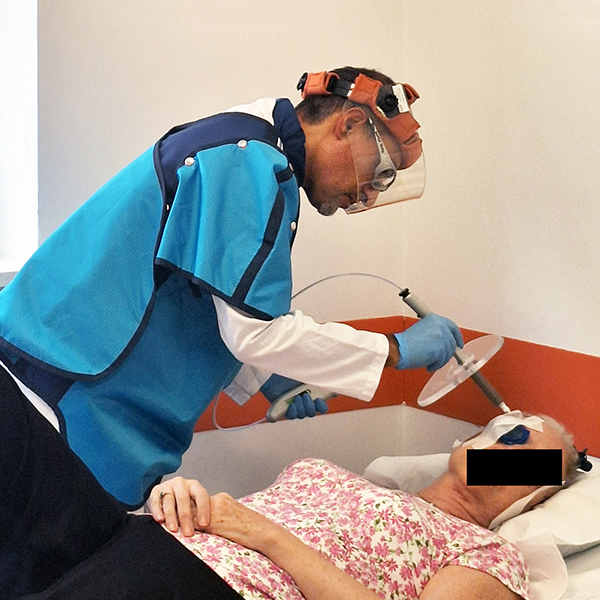
Rhenium skin cancer therapy is an advanced radionuclide therapy that offers a painless,*1,2 single-session,†1-3 non-invasive‡1 treatment without disfiguring scarring.1,4 Treatment with Rhenium-SCT employs a superficial application of a paste containing β-emitting particles directly over the NMSC lesion, targeting cancer cells without the need for surgery.1,4
Dr. Mohsen Farsad at Bolzano Hospital has been impressed with the Rhenium-SCT device, noting, "I have witnessed impressive outcomes with several patients treated here in Italy already, and am pleased with the design of the device technology."
"Rhenium skin cancer therapy fulfills many patients' desires for a non-surgical alternative for the treatment of NMSCs especially in relation to difficult localisations. I am pleased to continue treating suitable patients with Rhenium-SCT", adds Dr. Farsad.
Shannon D. Brown III, CCO at OncoBeta, says, "While surgical intervention remains the standard-of-care for NMSC treatment, it often leads to suboptimal outcomes. Rhenium-SCT offers a non-surgical alternative with a simple application directly to the area needed to treat, and allows patients to resume their daily activities with minimal disruption."
According to GLOBOCAN estimates, the number of new NMSC cases in Italy is projected to rise from 29,000 to 41,700 between 2022 and 2045.5 Epidemiological data for Europe shows an annual incidence rate of NMSCs at 129.3 cases per 100,000 men and 90.8 per 100,000 women. Basal cell carcinoma (BCC) accounts for approximately 15% of all diagnosed cancers in Italy, with an annual incidence of around 100 cases per 100,000 inhabitants.6
Dr. Gerhard Dahlhoff, Medical Director at OncoBeta, emphasised, "Rhenium-SCT enables targeted, non-invasive treatment of NMSCs while preserving adjacent healthy tissue. This patient-friendly approach underscores our commitment to enhancing treatment options for those affected by NMSCs."
More on illi News
Rhenium-SCT is currently available in Germany, Austria, Switzerland, Italy, Spain, United Kingdom, South Africa and Australia. Rhenium-SCT will be provided to patients at Bolzano Hospital by OncoBeta's distribution partner, DSD Pharma.
About the Rhenium-SCT® (Skin Cancer Therapy)
Non-melanoma skin cancer (NMSC) is the most common form of cancer in humans.1 The most common cause of NMSC is sun exposure, while other predisposing factors include genetic skin conditions and immunosuppressive diseases or treatments.1,4
The Rhenium-SCT is a painless,*1,2 single session,†1-3 non-invasive‡1 therapy that provides aesthetic results, even in cases otherwise considered difficult to treat.1,4,7 The Rhenium-SCT utilizes the radioisotope rhenium-188 in an epidermal application with optimal properties for the treatment of NMSCs (non-melanoma skin cancers). The Rhenium-SCT is a precise, personalized therapy that is only applied to the area needed to treat without affecting the healthy tissue.4,7 The specially designed device ensures the Rhenium-SCT compound never comes in direct contact with the patient's skin and the application is safe and simple for the applying physician. Most cases of NMSCs (basal cell carcinomas and squamous cell carcinomas) can be treated using the Rhenium-SCT in one single session. †1-4 Scar-free healing of the treated lesion area and the regeneration of healthy tissue occurs usually within a few weeks after treatment.1,7,8
About OncoBeta®
OncoBeta® with its headquarters located in Garching near Munich, Germany, is a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies. Since its foundation, OncoBeta has concentrated its efforts on the development, regulatory approval(s) and commercialization of the epidermal radioisotope therapy Rhenium-SCT® (Skin Cancer Therapy), targeting NMSCs. OncoBeta has perfected the customized application and device management system in conformity with all health, safety, and environmental protection regulatory standards.
Find out more about the Rhenium-SCT at www.oncobeta.com
Follow us on social media:
More on illi News
LinkedIn: https://www.linkedin.com/company/oncobeta-gmbh/
Facebook: https://www.facebook.com/oncobeta/
Instagram: https://www.instagram.com/oncobeta_gmbh/
About DSD Pharma
DSD Pharma is a distributor for diagnostics, theranostics and radiopharmaceutical/medical devices in the field of nuclear medicine and radiopharmaceuticals. DSD Pharma offers a product portfolio developed together at the forefront of research to its customers. This ensures the safety of latest technologies for the HCP and the best available diagnosis/theranosis/therapy for the patient.
Find out more about DSD Pharma at www.dsd-pharma.com
Forward-looking statements
This announcement includes forward-looking statements that involve risks, uncertainties and other factors, many of which are outside of OncoBeta's control and which could cause actual results to differ materially from the results discussed in the forward-looking statements. Forward-looking statements include statements concerning OncoBeta's plans, objectives, goals, future events, performance and/or other information that is not historical information. All such forward-looking statements are expressly qualified by these cautionary statements and any other cautionary statements which may accompany the forward-looking statements. OncoBeta undertakes no obligation to publicly update or revise forward-looking statements to reflect subsequent events or circumstances after the date made, except as required by law.
*No reported pain during procedure.1,2
†Complete tumour regression in 98.5% of lesions treated.3
‡A treatment is considered non-invasive when no cut or break in the skin is created.9
References
Rhenium skin cancer therapy is an advanced radionuclide therapy that offers a painless,*1,2 single-session,†1-3 non-invasive‡1 treatment without disfiguring scarring.1,4 Treatment with Rhenium-SCT employs a superficial application of a paste containing β-emitting particles directly over the NMSC lesion, targeting cancer cells without the need for surgery.1,4
Dr. Mohsen Farsad at Bolzano Hospital has been impressed with the Rhenium-SCT device, noting, "I have witnessed impressive outcomes with several patients treated here in Italy already, and am pleased with the design of the device technology."
"Rhenium skin cancer therapy fulfills many patients' desires for a non-surgical alternative for the treatment of NMSCs especially in relation to difficult localisations. I am pleased to continue treating suitable patients with Rhenium-SCT", adds Dr. Farsad.
Shannon D. Brown III, CCO at OncoBeta, says, "While surgical intervention remains the standard-of-care for NMSC treatment, it often leads to suboptimal outcomes. Rhenium-SCT offers a non-surgical alternative with a simple application directly to the area needed to treat, and allows patients to resume their daily activities with minimal disruption."
According to GLOBOCAN estimates, the number of new NMSC cases in Italy is projected to rise from 29,000 to 41,700 between 2022 and 2045.5 Epidemiological data for Europe shows an annual incidence rate of NMSCs at 129.3 cases per 100,000 men and 90.8 per 100,000 women. Basal cell carcinoma (BCC) accounts for approximately 15% of all diagnosed cancers in Italy, with an annual incidence of around 100 cases per 100,000 inhabitants.6
Dr. Gerhard Dahlhoff, Medical Director at OncoBeta, emphasised, "Rhenium-SCT enables targeted, non-invasive treatment of NMSCs while preserving adjacent healthy tissue. This patient-friendly approach underscores our commitment to enhancing treatment options for those affected by NMSCs."
More on illi News
- Naperville Police Participate in St. Patrick's Day Traffic Safety Campaign
- New partnership bringing trauma-informed libraries to vulnerable children
- Training Lofts Launches $1,099 Unlimited Training Membership Featuring Semi-Private Coaching, Nutrition Support, and Recovery Services
- Naperville and Aurora Joint Release: North Aurora Road Closure between Pennsbury Lane & Frontenac Road Starts March 31
- American Properties Realty, Inc. Leadership Attends NAHB International Builders' Show in Florida
Rhenium-SCT is currently available in Germany, Austria, Switzerland, Italy, Spain, United Kingdom, South Africa and Australia. Rhenium-SCT will be provided to patients at Bolzano Hospital by OncoBeta's distribution partner, DSD Pharma.
About the Rhenium-SCT® (Skin Cancer Therapy)
Non-melanoma skin cancer (NMSC) is the most common form of cancer in humans.1 The most common cause of NMSC is sun exposure, while other predisposing factors include genetic skin conditions and immunosuppressive diseases or treatments.1,4
The Rhenium-SCT is a painless,*1,2 single session,†1-3 non-invasive‡1 therapy that provides aesthetic results, even in cases otherwise considered difficult to treat.1,4,7 The Rhenium-SCT utilizes the radioisotope rhenium-188 in an epidermal application with optimal properties for the treatment of NMSCs (non-melanoma skin cancers). The Rhenium-SCT is a precise, personalized therapy that is only applied to the area needed to treat without affecting the healthy tissue.4,7 The specially designed device ensures the Rhenium-SCT compound never comes in direct contact with the patient's skin and the application is safe and simple for the applying physician. Most cases of NMSCs (basal cell carcinomas and squamous cell carcinomas) can be treated using the Rhenium-SCT in one single session. †1-4 Scar-free healing of the treated lesion area and the regeneration of healthy tissue occurs usually within a few weeks after treatment.1,7,8
About OncoBeta®
OncoBeta® with its headquarters located in Garching near Munich, Germany, is a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies. Since its foundation, OncoBeta has concentrated its efforts on the development, regulatory approval(s) and commercialization of the epidermal radioisotope therapy Rhenium-SCT® (Skin Cancer Therapy), targeting NMSCs. OncoBeta has perfected the customized application and device management system in conformity with all health, safety, and environmental protection regulatory standards.
Find out more about the Rhenium-SCT at www.oncobeta.com
Follow us on social media:
More on illi News
- Mayor Brandon Johnson Announces 2nd Annual Chicago River Swim Following Historic First Year, Establishing New Civic Tradition
- $317M Revenue and a Clear Path to $1B: $IQST is Positioned for a Major Profitability Inflection
- Jones Software Corp Expands AI Skilling Through Microsoft Commercial Marketplace with Global Deliver
- ASI Hosts 2026 Executive Business Summit for Global Partner Community
- Pastor Saeed Abedini Releases THE TRUTH – Volume 1, A Deeply Personal Story of Faith, Struggle, and Redemption
LinkedIn: https://www.linkedin.com/company/oncobeta-gmbh/
Facebook: https://www.facebook.com/oncobeta/
Instagram: https://www.instagram.com/oncobeta_gmbh/
About DSD Pharma
DSD Pharma is a distributor for diagnostics, theranostics and radiopharmaceutical/medical devices in the field of nuclear medicine and radiopharmaceuticals. DSD Pharma offers a product portfolio developed together at the forefront of research to its customers. This ensures the safety of latest technologies for the HCP and the best available diagnosis/theranosis/therapy for the patient.
Find out more about DSD Pharma at www.dsd-pharma.com
Forward-looking statements
This announcement includes forward-looking statements that involve risks, uncertainties and other factors, many of which are outside of OncoBeta's control and which could cause actual results to differ materially from the results discussed in the forward-looking statements. Forward-looking statements include statements concerning OncoBeta's plans, objectives, goals, future events, performance and/or other information that is not historical information. All such forward-looking statements are expressly qualified by these cautionary statements and any other cautionary statements which may accompany the forward-looking statements. OncoBeta undertakes no obligation to publicly update or revise forward-looking statements to reflect subsequent events or circumstances after the date made, except as required by law.
*No reported pain during procedure.1,2
†Complete tumour regression in 98.5% of lesions treated.3
‡A treatment is considered non-invasive when no cut or break in the skin is created.9
References
- Castellucci P, et al. Eur J Nucl Med Mol Imaging. 2021; 48: 1511–1521.
- Cipriani C, et al. J Dermatol Treat. 2022;33(2):969–975. Epub 22 Jul 2020.
- Cipriani C, et al. In Therapeutic Nuclear Medicine. 2014. RP Baum (Ed), New York: Springer.
- Tietze JK, et al. Clin Nucl Med. 2023;48:869–76.
- Global Burden of Disease Cancer Collaboration, et al. JAMA Oncol. 2019;5(12):1749-1768.
- Sordi E, et al. Epidemiologia. 2024;5:1–10.
- Cipriani C, et al. International J Nucl Med. 2017; July:114–112.
- Sedda AF, et al. Clin Exp Dermatol. 2008;33(6):745–749.
- Australian Therapeutic Goods Administration. Acronym and glossary of terms. Available at: https://www.tga.gov.au/resources/acronyms-and-glossary-terms. (Accessed June 2024).
Source: OncoBeta GmbH
0 Comments
Latest on illi News
- Buildout Launches CRM, Completing the Industry's First AI-Powered End-to-End Deal Engine for CRE
- The Franchise King® Releases Free Guide for Nervous Buyers
- Kanguro Insurance Taps Paylode to Launch Best-in-Class Pet and Renters Insurance Rewards Experience
- CCHR: CIA Mind-Control Files Raise Urgent Questions as Millions Take Psychotropic Drugs
- NRx Pharmaceuticals Launches Breakthrough One-Day Treatment Clinic in Florida as FDA Pathway and Clinical Data Strengthen Growth Outlook; $NRXP
- Revenue Optics Launches Talent Infrastructure Platform for SaaS Revenue Hiring and Appoints Sabz Kaur to Lead Growth
- Building a Multi-Domain Autonomous Systems Platform at the Intersection of AI, Defense and Infrastructure: VisionWave Holdings (N A S D A Q: VWAV)
- Bent Danholm Named "Top Luxury Real Estate Leader" in Modern Luxury Miami
- Author Ken Mora to Celebrate New Caravaggio Book Debut with Special Event at Palazzo Venezia Naples
- The Friendly Music Community Unveils The Friendly Listening Room with Premier Performance by Acclai
- Matthew Sisneros Releases Raw and Unfiltered Memoir: The Devil Lost Another One — A Powerful Story of Crime, Consequence, and Redemption
- From Life to Light: Jess L. Martinez Shares a Soulful Poetry Collection That Explores What It Means to Be Human
- Lawsuit Filed Against Boeing Over Defective Seat Switch on Boeing 787
- New Portable Toilet Drain Adapter Eliminates Manual Waste Tank Emptying
- Quadcode Acquires Significant Stake in Game 7, LLC - The Parent Company for FPFX Tech and PropAccount.com
- Night Shade Introduces Smart Privacy Protection for Modern Devices
- Innovative Ear Muff Hat Combines Warmth and Convenience in One Winter Accessory
- Danholm Collection Announces Sale of 16689 Broadwater Ave in Winter Garden, Highlighting Strong Performance in Twinwaters Community
- New Repositionable Handheld Easel Offers Artists Greater Freedom and Flexibility
- Strong Clinical Results for Breakthrough Liver Diagnostic Platform; ENDRA Life Sciences (N A S D A Q: NDRA) $NDRA