Trending...
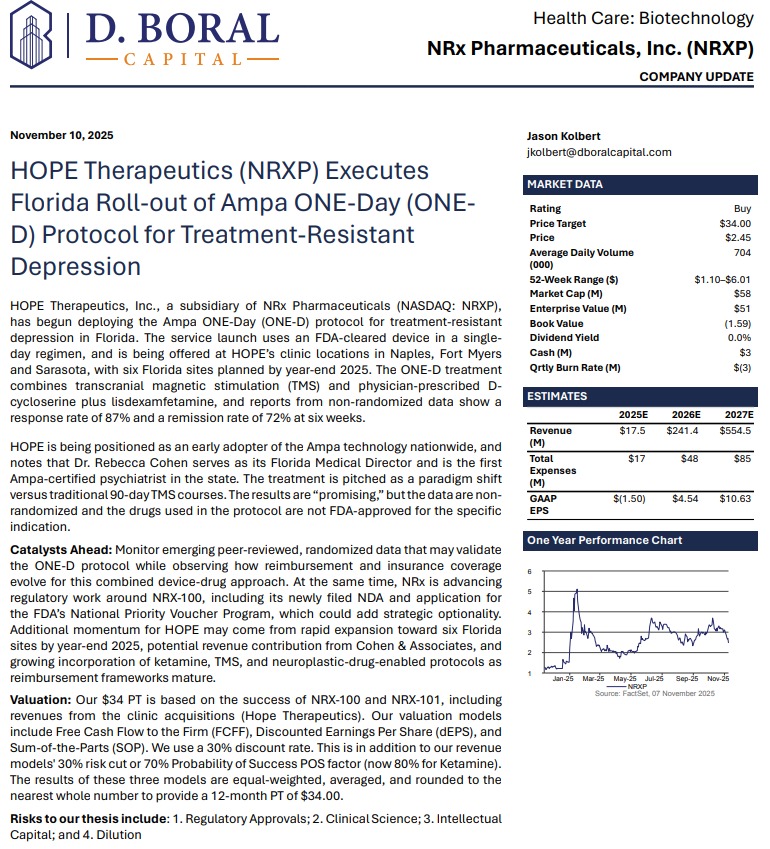
- Greater Englewood Chamber's Solar + EV Training Program Returns for Green Energy Careers
- People & Stories/Gente y Cuentos Welcomes Two New Trustees as Organization Enters 54th Year and Expands Community Reach
- Mel Blackwell to Keynote 2026 NSSF Marketing and Leadership Summit
NRx Pharmaceuticals (N A S D A Q: NRXP) $NRXP: Dual FDA Pathways, and an Expanding AI-Driven Treatment Ecosystem Position NRXP at the Forefront of a Multi-Billion-Dollar Mental Health Transformation
MIAMI - illiNews -- NRx Pharmaceuticals (N A S D A Q: NRXP) is rapidly emerging as a high-impact, late-stage biotech story transitioning into a nationally aligned healthcare platform, as a powerful convergence of regulatory clarity, federal policy support, and technological innovation reshapes both its timeline and total addressable market.
The company's latest developments place it squarely at the intersection of urgent public health need and government-backed acceleration, following the April 18, 2026 Presidential Executive Order aimed at fast-tracking treatments for serious mental illness, including psychedelic and breakthrough therapies.
This directive is expected to expand the use of real-world evidence, accelerate FDA review pathways, and introduce priority mechanisms such as Commissioner's National Priority Vouchers—one of which NRx has already applied for. The result is a meaningful shift in the regulatory landscape, positioning companies like NRx to benefit from faster approvals, increased prioritization, and national-level visibility.
FDA Alignment Transforms the Timeline
At the core of the investment thesis is a rare and highly material regulatory breakthrough. Following direct engagement with the FDA, NRx confirmed that its lead candidate, NRX-100 (preservative-free ketamine), can proceed toward a New Drug Application using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This positions the company to target an NDA submission as early as June 2026 while simultaneously expanding the proposed indication to include a broader population of patients suffering from treatment-resistant depression with suicidality.
To support this filing, NRx is preparing to submit data from more than 70,000 patients, creating one of the most robust real-world datasets in the psychiatric treatment space.
Key regulatory positioning now includes:
Dual FDA Catalysts Create a Rare Parallel Opportunity
More on illi News
While the NDA pathway represents transformational upside, NRx is simultaneously advancing a second regulatory track that may generate nearer-term revenue.
The company's Abbreviated New Drug Application for preservative-free ketamine in anesthesia has already achieved bioequivalence confirmation and near-final labeling alignment, with anticipated FDA action in the second half of 2026.
This dual-track structure creates a compelling dynamic:
Few companies at this stage possess two aligned regulatory catalysts with overlapping timelines.
From Biotech to Platform: Expanding Into Neurotechnology and AI
NRx is no longer operating as a single-asset biotech. It is building an integrated platform that combines pharmaceuticals, neurostimulation, and artificial intelligence.
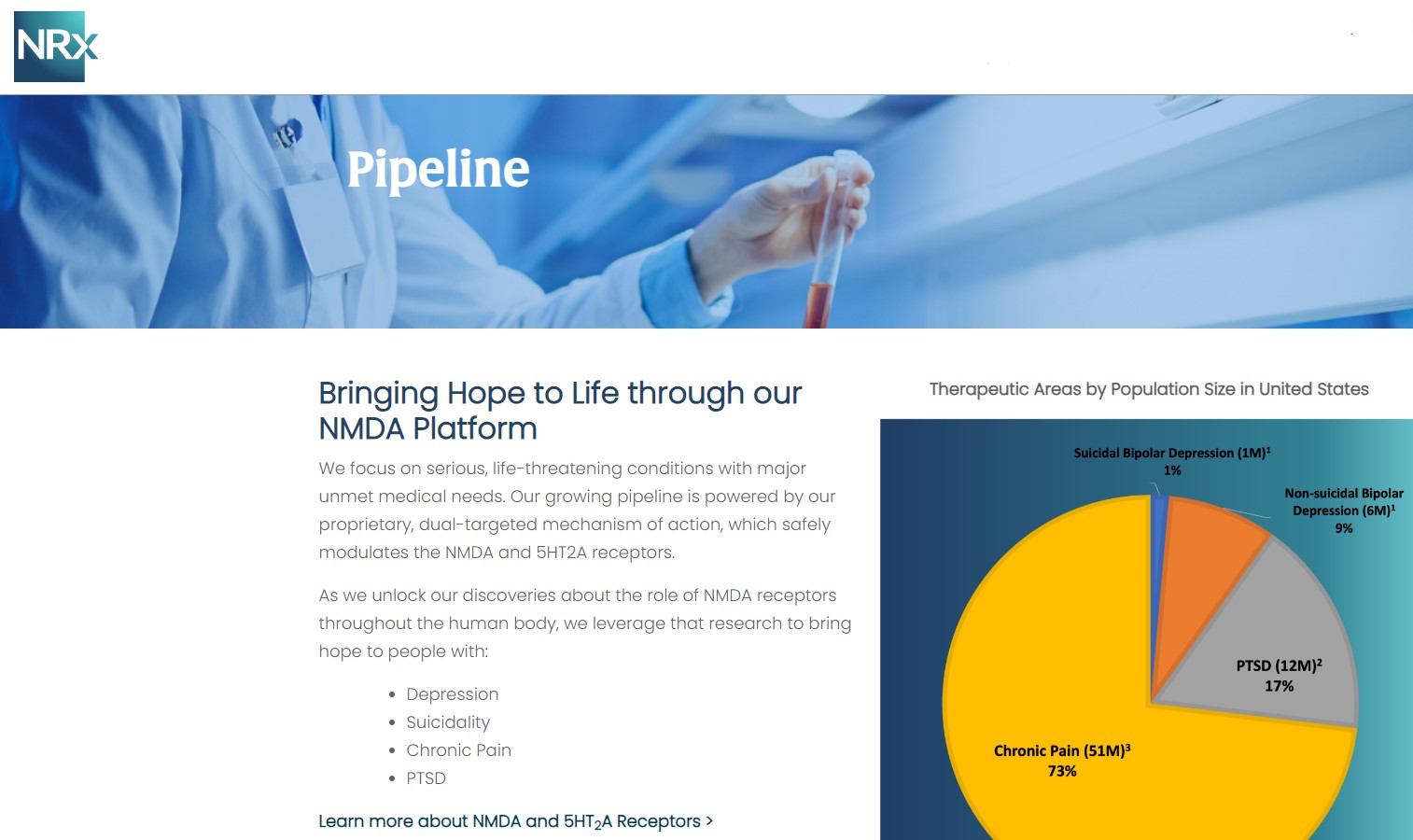
Its Breakthrough Therapy-designated candidate, NRX-101, is being developed as a maintenance therapy for suicidal bipolar depression and chronic pain, designed to work synergistically with neuroplastic treatment approaches.
That strategy is now expanding into advanced neurotechnology through the formation of NRx Defense Systems, a subsidiary focused on robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy.
This initiative targets military personnel and first responders—populations with significantly elevated rates of PTSD and depression—and introduces the potential for government-backed, non-dilutive funding channels.
Technology and platform expansion highlights:
Commercial Infrastructure Already in Motion
Importantly, NRx is not waiting for regulatory approvals to build its business. Through its HOPE Therapeutics subsidiary, the company has established a growing network of interventional psychiatry clinics delivering neuroplastic therapies, including ketamine and TMS.
These clinics are already generating revenue and are supported by a combination of government programs, insurance reimbursement, and private-pay patients.
In parallel, the company has partnered with neurocare AG to expand a nationwide treatment network that includes:
This infrastructure provides a ready-made commercialization channel upon approval.
More on illi News
AI Integration Adds a New Layer of Precision Medicine
NRx is further differentiating itself through the integration of artificial intelligence into patient care.
Through its partnership with Emobot, the company is deploying a passive monitoring platform—often described as a "Depression Thermometer"—that continuously evaluates patient condition via smartphone-based data collection.
This enables earlier intervention, improved treatment optimization, and a scalable model for managing chronic mental health conditions.
Financial Position Strengthens Execution Capability
The company has also made meaningful progress in strengthening its financial foundation.
Recent disclosures highlight reduced operating expenses, the elimination of balance sheet debt, and sufficient capital to support operations through 2026.
This positions NRx to execute on its regulatory and commercial strategy without immediate financing pressure.
A Market Defined by Urgency and Scale
NRx is targeting one of the most significant unmet needs in healthcare. More than 13 million Americans report seriously considering suicide each year, while treatment-resistant depression and PTSD continue to rise globally.
With ketamine-based therapies demonstrating rapid clinical impact and federal policy now accelerating access to innovative treatments, the demand environment is both immediate and expanding.
Final Take: A Convergence Story with Multiple Inflection Points
NRx Pharmaceuticals is no longer simply advancing a drug candidate—it is building a multi-dimensional platform at the intersection of biotech, AI, neurotechnology, and national healthcare policy.
With:
NRx now represents a rare type of opportunity in the market:
A company where regulatory clarity, government support, and commercial execution are all advancing at the same time
For aggressive, forward-looking investors, NRXP is not just approaching a catalyst—
$NRXP is entering a phase where multiple catalysts are beginning to compound.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
The company's latest developments place it squarely at the intersection of urgent public health need and government-backed acceleration, following the April 18, 2026 Presidential Executive Order aimed at fast-tracking treatments for serious mental illness, including psychedelic and breakthrough therapies.
This directive is expected to expand the use of real-world evidence, accelerate FDA review pathways, and introduce priority mechanisms such as Commissioner's National Priority Vouchers—one of which NRx has already applied for. The result is a meaningful shift in the regulatory landscape, positioning companies like NRx to benefit from faster approvals, increased prioritization, and national-level visibility.
FDA Alignment Transforms the Timeline
At the core of the investment thesis is a rare and highly material regulatory breakthrough. Following direct engagement with the FDA, NRx confirmed that its lead candidate, NRX-100 (preservative-free ketamine), can proceed toward a New Drug Application using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This positions the company to target an NDA submission as early as June 2026 while simultaneously expanding the proposed indication to include a broader population of patients suffering from treatment-resistant depression with suicidality.
To support this filing, NRx is preparing to submit data from more than 70,000 patients, creating one of the most robust real-world datasets in the psychiatric treatment space.
Key regulatory positioning now includes:
- NDA pathway for NRX-100 with no additional trials required
- Fast Track designation already in place
- Real-world evidence accepted as part of the approval framework
- Expanded label strategy significantly increasing market potential
Dual FDA Catalysts Create a Rare Parallel Opportunity
More on illi News
- AI Is Closing the Gap Between Offshore Virtual Assistants and Onshore Staff
- Naperville: 'Click It or Ticket' campaign underway for Memorial Day
- CCHR Highlights Concerns Over Coercive and Failed $140 Billion Mental Health Practices at Psychiatric Convention
- Avery Headley Leads Major Stabilization and Modernization Initiative Across Bronx Affordable Housing Portfolio
- NewReputation's AI Sentiment Analysis Tool Reaches 2,500 Users as Businesses Demand Clearer Brand Intelligence
While the NDA pathway represents transformational upside, NRx is simultaneously advancing a second regulatory track that may generate nearer-term revenue.
The company's Abbreviated New Drug Application for preservative-free ketamine in anesthesia has already achieved bioequivalence confirmation and near-final labeling alignment, with anticipated FDA action in the second half of 2026.
This dual-track structure creates a compelling dynamic:
- Near-term commercialization potential via ANDA approval
- Longer-term, high-value expansion via NDA approval
Few companies at this stage possess two aligned regulatory catalysts with overlapping timelines.
From Biotech to Platform: Expanding Into Neurotechnology and AI
NRx is no longer operating as a single-asset biotech. It is building an integrated platform that combines pharmaceuticals, neurostimulation, and artificial intelligence.
Its Breakthrough Therapy-designated candidate, NRX-101, is being developed as a maintenance therapy for suicidal bipolar depression and chronic pain, designed to work synergistically with neuroplastic treatment approaches.
That strategy is now expanding into advanced neurotechnology through the formation of NRx Defense Systems, a subsidiary focused on robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy.
This initiative targets military personnel and first responders—populations with significantly elevated rates of PTSD and depression—and introduces the potential for government-backed, non-dilutive funding channels.
Technology and platform expansion highlights:
- Development of robotic, AI-guided TMS systems
- Integration with NRX-101 for enhanced neuroplastic response
- Prototype expected mid-2026
- Clinical data showing up to 87% response rates in related therapies
Commercial Infrastructure Already in Motion
Importantly, NRx is not waiting for regulatory approvals to build its business. Through its HOPE Therapeutics subsidiary, the company has established a growing network of interventional psychiatry clinics delivering neuroplastic therapies, including ketamine and TMS.
These clinics are already generating revenue and are supported by a combination of government programs, insurance reimbursement, and private-pay patients.
In parallel, the company has partnered with neurocare AG to expand a nationwide treatment network that includes:
- 20+ clinical sites
- More than 400 installed TMS systems
This infrastructure provides a ready-made commercialization channel upon approval.
More on illi News
- CAPO Supply Announces Opening of Second Location in New Castle, Pennsylvania
- Wheaton College Honors Kharis Publishing Founder with 2026 Presidential Award
- $224 Billion Growing Market in Life Settlements Presents Major Opportunity for New Policy Acquisition Business Plan: DLT Resolution Stock Symbol: DLTI
- Fyt-02 Launches on Kickstarter The Smart Sensor That Turns Any Chair Into a Posture & Movement Track
- YieldOMega Launches $DOUB Airdrop Campaign Ahead of TimeCurve Launch
AI Integration Adds a New Layer of Precision Medicine
NRx is further differentiating itself through the integration of artificial intelligence into patient care.
Through its partnership with Emobot, the company is deploying a passive monitoring platform—often described as a "Depression Thermometer"—that continuously evaluates patient condition via smartphone-based data collection.
This enables earlier intervention, improved treatment optimization, and a scalable model for managing chronic mental health conditions.
Financial Position Strengthens Execution Capability
The company has also made meaningful progress in strengthening its financial foundation.
Recent disclosures highlight reduced operating expenses, the elimination of balance sheet debt, and sufficient capital to support operations through 2026.
This positions NRx to execute on its regulatory and commercial strategy without immediate financing pressure.
A Market Defined by Urgency and Scale
NRx is targeting one of the most significant unmet needs in healthcare. More than 13 million Americans report seriously considering suicide each year, while treatment-resistant depression and PTSD continue to rise globally.
With ketamine-based therapies demonstrating rapid clinical impact and federal policy now accelerating access to innovative treatments, the demand environment is both immediate and expanding.
Final Take: A Convergence Story with Multiple Inflection Points
NRx Pharmaceuticals is no longer simply advancing a drug candidate—it is building a multi-dimensional platform at the intersection of biotech, AI, neurotechnology, and national healthcare policy.
With:
- Federal acceleration of mental health treatment approvals
- FDA-aligned NDA pathway requiring no new trials
- Parallel ANDA approval opportunity
- Breakthrough Therapy designation for NRX-101
- Expanding clinic network generating real-world revenue
- AI-driven patient monitoring and treatment optimization
- Entry into defense-focused neurotechnology
NRx now represents a rare type of opportunity in the market:
A company where regulatory clarity, government support, and commercial execution are all advancing at the same time
For aggressive, forward-looking investors, NRXP is not just approaching a catalyst—
$NRXP is entering a phase where multiple catalysts are beginning to compound.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on illi News
- iatroX surpasses 500,000 clinical queries and expands specialist exam coverage
- Inside-Out Hollywood: The Relentless Rise of Joseph Nybyk (AKA Joseph Neibich)
- SRK Collective Media Group Launches with a Modern Approach to Media, Authority Building, and Cultural Visibility
- MSBG Corporation Acquires GridWatch US Telemetry Automation System
- TAYP Expands Athlete Exposure Platform Beyond Georgia With New Push Into Virginia and the 757
- KT Medical Staffing Expands Concierge Nursing and Private Duty Nursing Services in Orange County
- Mayor Brandon Johnson, Department of Planning and Development Announce Neighborhood Opportunity Fund Grants For 11 Chicago Businesses
- The Millennium Alliance Achieves Great Place To Work® Certification™ Amid Continued Growth
- The Millennium Alliance Appoints Former Adweek Executive Eric Hayden Shakun as Chief Financial Officer to Accelerate Next Phase of Growth
- T. Jones Group Named Finalist Across Multiple Categories at the 2026 Georgie Awards
- The Simplest Small Business You're Probably Not Thinking About
- San Francisco Writer Wins Webby Award, Internet's Highest Honor, for Website Based on her Novel
- EDC Weekend Comedy Special Featuring Don Barnhart & Friends — Use Promo Code FRIEND for 50% Off
- N Y S E: OTH Off The Hook YS Is Building a Vertically Integrated Marine Empire — And Investors Are Starting to Notice
- Concierge Title Agency Merges with Independence Title, Inc. to Deliver an Expanded Concierge Closing Experience Across South Florida
- Grow My Security Company Launches Next-Generation Website and Expands Strategic Marketing Solutions for the Security Industry
- $4.8M in Contracted AI Revenue with Projections of $30M Over 6-12 Months for Diversified AI Software and Platform-Based Services Provider XMax Inc
- Michelangelo's Great Secret Hiding in Plain Sight
- Virginia Marchese's Paradox: A Nation Still Deciding Who Belongs Examines Race, Migration, Law, and America's Unfinished Struggle for Equality
- From Blank Page to Published Book