Trending...
- NEW MANAGEMENT BOOK: Creating a Joy-Centric Culture
- KeysCaribbean Offers 'Skip-the-Crowds' Savings With 15 Percent Off April Stays
- Red5 Taps PubNub to Power the Next Era of Real-Time Interactive Streaming
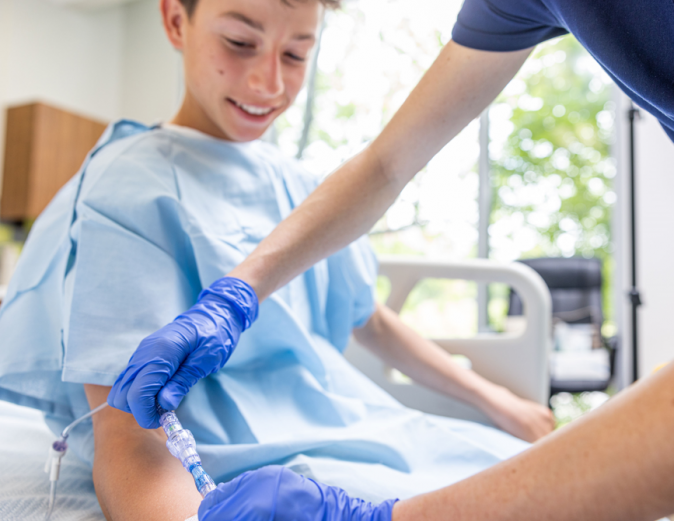
FAYETTEVILLE, Ark. - illiNews -- Lineus Medical, a medical device company focused on reducing IV complications, announced that the company's flagship product, SafeBreak Vascular, has received clearance from the U.S. Food and Drug Administration (FDA) for use in pediatric patients. SafeBreak Vascular has been shown in adult clinical studies to reduce IV complications with its innovative breakaway technology1.
SafeBreak Vascular is the first breakaway device cleared by the FDA and proven to reduce IV complications in adults by 44%1. It fits between the IV tubing from the IV pump and the patient's IV access point at the arm. When there is a damaging force on the line, SafeBreak separates to protect the patient's IV. This innovative feature helps prevent accidental dislodgement of the catheter, as well as other IV complications caused by damaging forces on the IV line1. This is especially important for pediatric patients who may be more active, less inclined to follow instructions, and less aware of their surroundings.
More on illi News
"We are incredibly proud to receive clearance from the FDA for the use of SafeBreak Vascular with pediatric patients," said Vance Clement, CEO of Lineus Medical. "SafeBreak provides a simple, effective solution for preventing IV complications, which reduces IV restarts and additional needlesticks for patients. Now, it doesn't matter if the patient is 1 or 100 years old, SafeBreak is cleared for use on all peripheral IVs. Our goal is to have a SafeBreak on every IV line, everywhere in the world, and this additional clearance lets us help one of the most vulnerable populations that needs it the most, our kids."
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical tubing may be reduced as breakaway devices separate to protect the patient. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Twitter or LinkedIn.
1 Dipper Study Data on File
MKG-0194 REV 00
SafeBreak Vascular is the first breakaway device cleared by the FDA and proven to reduce IV complications in adults by 44%1. It fits between the IV tubing from the IV pump and the patient's IV access point at the arm. When there is a damaging force on the line, SafeBreak separates to protect the patient's IV. This innovative feature helps prevent accidental dislodgement of the catheter, as well as other IV complications caused by damaging forces on the IV line1. This is especially important for pediatric patients who may be more active, less inclined to follow instructions, and less aware of their surroundings.
More on illi News
- Homeowner Prep Announces Strategic Language Shift: Replacing "Renters" with "Future Homeowners" to Inspire Wealth-Building Mindsets
- LiposoMore™ Redefines Bioavailability: Joyful Nutritional Launches High-Performance Liposomal Vitamin C and Iron for the Global Supplement Market
- GDE Tree Services Expands Operations into Sydney, NSW
- Tuckwell Machinery Expands CNC Range to Support Australian Cabinet Makers
- The Inner Power of Emotional Self-Leadership
"We are incredibly proud to receive clearance from the FDA for the use of SafeBreak Vascular with pediatric patients," said Vance Clement, CEO of Lineus Medical. "SafeBreak provides a simple, effective solution for preventing IV complications, which reduces IV restarts and additional needlesticks for patients. Now, it doesn't matter if the patient is 1 or 100 years old, SafeBreak is cleared for use on all peripheral IVs. Our goal is to have a SafeBreak on every IV line, everywhere in the world, and this additional clearance lets us help one of the most vulnerable populations that needs it the most, our kids."
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a unique, patented, break-away technology that is designed to work with all types of medical tubing. Complications with feeding tubes, IV lines, urinary catheters and any other type of medical tubing may be reduced as breakaway devices separate to protect the patient. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Twitter or LinkedIn.
1 Dipper Study Data on File
MKG-0194 REV 00
Source: Lineus Medical
Filed Under: Health, Technology
0 Comments
Latest on illi News
- Chicago: Mayor Brandon Johnson and BACP Announce Selected Organizations for the Neighborhood Retail Activation Program
- TicTac Group acquires French EdTech company Distrisoft
- Suburban Orthopaedics Expands Comprehensive Orthopaedic Care in Northwest Suburbs
- Chicago: Mayor Brandon Johnson Joins Goodman Theatre To Celebrate Opening of Theater of The Mind
- Chicago: Mayor Brandon Johnson Statement on Preliminary Ruling Restoring Red Line Extension Funding
- Kettera Strategies Selects Bite Stream
- Mark Dobosz Makes Donorassess.org Free To Every Nonprofit On The Planet
- Genpak Announces Closure of Utah Manufacturing Facility
- Vanguard Gifted Academy Students Join Millions Globally for World Maths Day 2026
- Newborn Care Network Introduces Clinical Standard to Bridge the Six-Week Postpartum Gap
- The AAA Metamorphosis: How Global Gaming Is Redefining Production Standards
- Monexplora Explains the Options Mechanics Behind March's Tech Selloff and VIX Surge
- Ill House District 83, Candidate, Loyola Grad Calls for Corrective Action Due to Gorman Murder
- Larry R. Wasion Highlights Jump Gate I: Time Chair. The Opening Novel in His Expansive Science Fiction Series
- Mayor Brandon Johnson Launches Repair Chicago Engagement Series and Survey to Inform City's Reparations Study
- New Book Reveals The Science Of Predictions
- Compliance Alert: Maryland, Texas Regulate Use of Artificial Intelligence in Utilization Reviews
- Elite Benefits Launches "Benefits Survival Guide" to Help Employers Control Costs and Uncertainty
- Colony Ridge Communities Celebrates Successful Soccer Season Kickoff with Families and Youth
- Pregis Launches Sharp™ Lynx 18 Automated Bagger: Unlocking Packing Efficiency in Space-Constrained Fulfillment Operations